 
As a result, B’s electron cloud may be split into three halves. The B atom in BF3 is surrounded by electron domains, but how many are there? The number of atoms of F that are covalently linked to B is 3. There are three distinct regions where electrons may exist. No lonely hearts Since the atoms are all clustered together in four groups, no unpaired atoms are present. The oxygen atoms in CO2 have two unpaired electrons each. The number of lone pairs in CO2’s Lewis structure. Separated into two separate pairs H2O, AB2E2, Water There are four possible bonding configurations in a water molecule: (see figure below). In order to predict a geometry, electrons are distributed around a core metal atom in such a way as to minimise and lessen the greatest repulsion between another atom. The electron domain is used to anticipate the shape of the molecule by counting the number of lone pairs and bonds around the core metal atom. The molecular geometry of NH3 is that of a trigonal pyramid, with the three Hydrogen atoms placed symmetrically around the Nitrogen atom at the base and the two nonbonding electrons at the top. What is NH3 electron geometry?Ĭhemical Structure of NH3 The molecular structure of ammonia is tetrahedral. One can only have a linear molecular geometry in the linear domain. The atom A in the molecule’s core is a quadruple-domain atom. When two atoms form a connection, all of their shared electrons are on the same side of the core atom, making them a single electron domain.  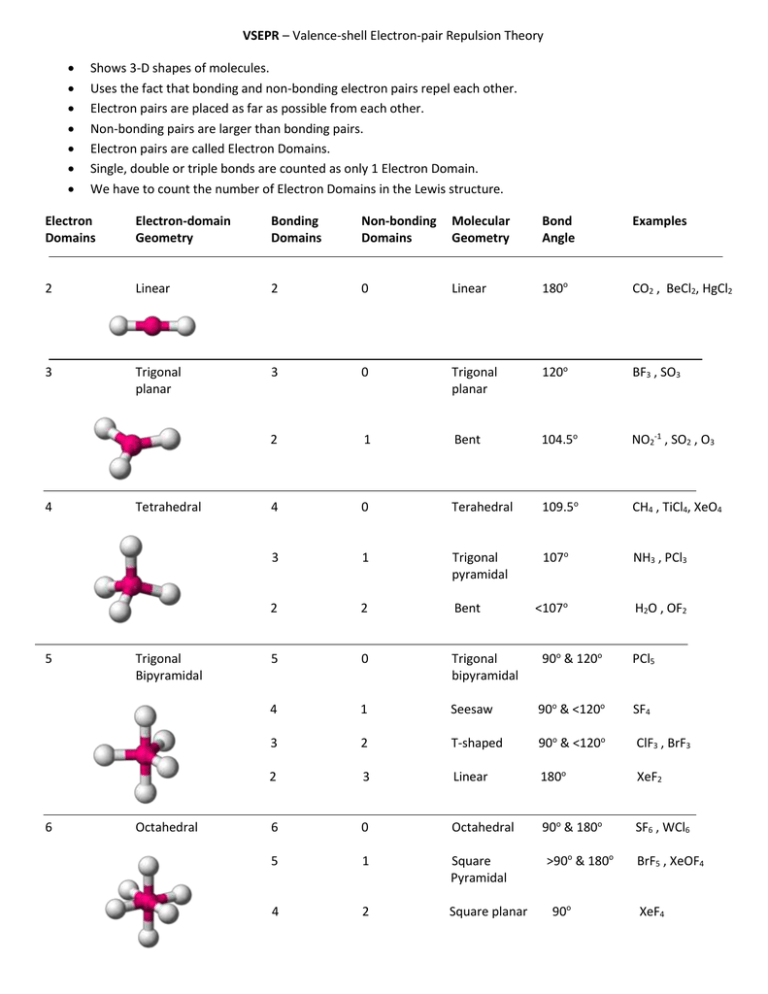
How many electron domains does A linear bond have? Each noncentral atom counts as one electron domain on the central atom, regardless of whether the bonded atom is attached with a single, double, or triple bond. There are 4 different regions where electrons may exist Structure of Atoms with Three to Six Electron Domains Domains of Electrons and Their Typical Organization 2 CO2 Linear Triangles and a Plane Make a (3 electron domains) (BrF3, AlCl3, SnCl2) In the form of a tetrahedron, with 4 faces (4 electron domains) The Chemical Formulas for CH4 and SiCl4 How many electron domains are in A single bond?Ī bound atom and a nonponding pair of electrons are examples of electron domains. There are a total of eight pairs of electrons, and the four that determine form have a tetrahedral geometry. (4) pairs of electrons Both ammonia and nitrogen have four electron pairs, and nitrogen’s coordination geometry is based on a tetrahedral arrangement of electron pairs. Molecular Name Dubbed “the silent killer,” carbon dioxide (CO2) Nature Substance that does not interact with the magnetic field Co2 has 16 valence electrons and no formal charge. All the oxygen atoms have two lone pairs. As many as four isolated pairs may be seen in the lewis structure of CO2. How many electron domains does CO2 have?Ĭarbon dioxide has a linear molecular geometry. The steric number is another name for the electron domain. Two solitary bonds and two lone pairs that don’t contribute to bonds make up water’s four regions. One domain of electrons is exemplified by a lone pair, single, double, or triple bond. “Electron Domain ô” The probable place to find electrons (bonding and nonbonding). What is the electron domain geometry of bf3?.What is the electron domain geometry of SF4?.How many electron domains does A linear bond have?.How many electron domains are in A single bond?.How many electron domains does CO2 have?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
